Abstract
Introduction - Rising NPM1 mutant minimal residual disease (MRD) is strongly associated with relapse post-chemotherapy for Acute Myeloid Leukaemia (AML). Utility of other mutations affecting DNMT3A, IDH1 and IDH2 for MRD tracking has been hampered by persistence of clonal hematopoiesis after chemotherapy. In the post-allogeneic stem cell transplant (alloHSCT) context however, we show that host-derived clonal hematopoiesis is erased by donor hematopoiesis. We therefore sought to examine kinetics of re-emergence of host-derived mutations in CD34+, CD33+ CD3+ fractionated peripheral blood (PB) and correlation with loss of donor chimerism (DC) and clinical outcome.
Aim - To utilize ddPCR monitoring to track mutant DNMT3A, IDH1, IDH2 and NPM1 clones in CD34+/CD33+ and CD3+ sorted peripheral blood fractions following alloHSCT for AML.
Methods - Patients undergoing alloHSCT between January 2014 and December 2016 with known mutations in the hotspot regions of DNMT3A, IDH1, IDH2 and NPM1 as detected by Sequenom Mass Array were included in the analysis cohort. An unfractionated pre-transplant peripheral blood sample was utilized for mutation determination, with serial fractionated PB samples analyzed and stored as part of routine DC assessment. DC was assessed by selection of disparate donor-recipient short tandem repeats (STR) sequences. CD3+, CD33+ CD34+ cell fractions sorted using MAC beads. Cell fractions with sufficient input DNA were analyzed by ddPCR utilizing FAM and HEX probes to quantify mutant and wildtype DNMT3A R882H and R882C, IDH1 R132C and R132H, IDH2 R140Q and NPM1 insA. Mutant and wild-type copy number was determined utilizing Poisson statistics and fractional abundance (FA) determined. Statistical analysis was performed in R version 3.3.2 and GraphPad Prism version 7.0b.
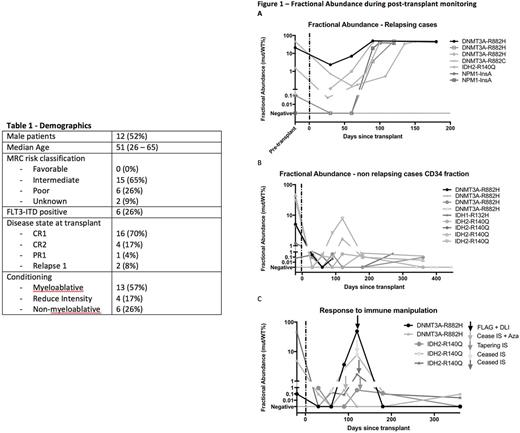
Results - 23 patients were analyzed with 9 with DNMT3A (8 R882H, 1 R882C), 2 IDH1 (1 R132C, 1 R132H), 6 IDH2 (all R140Q) and 14 NPM1 insA mutations. Demographic information is summarized in table 1. Based on known mutational profile of the cohort, FA of pre-transplant DNMT3A and IDH mutations ranged between 0.06% - 45.8%, while only 1 of 7 pre-transplant NPM1 samples was positive (0.0512%). FA rose earlier in CD34 fraction, then in CD33 or CD3 fractions. In 9 out of 10 paired pre-transplant and post-transplant samples, there was a significant reduction in FA (median 97% reduction), despite a variety of conditioning intensities. Many positive post-transplant blood specimens represented pre-leukemic mutations, with mutant positivity in both CD34+ and CD3+ fractions. In 6/6 cases with either a persistently high or rising peripheral blood mutant FA, clinical relapse occurred (Figure 1A). All 5 subjects with a progressive rise in mutant burden had detectable disease prior to clinical disease relapse by a median of 30 days (range 25 - 51 days). Low-level (<1%) fluctuating positivity was seen in most post-transplant cases (figure 1B). A single case demonstrated autologous recovery post-transplant with their IDH2 clone followed by relapse. Falling CD34+ chimerism to ≤80% was detected in 9 patients during surveillance. Of these, an emergent mutant clone was detected in all but one who was confirmed negative for NPM1 insA on RT-qPCR on marrow. In contrast to the pre-transplant setting, rising levels of rising levels of pre-leukemic IDH and DNMT3A from a negative baseline post donor engraftment was a strong predictor of impending relapse (figure 1A). 5 cases with emerging clones identified during MRD surveillance molecularly responded to immune manipulation, such as withdrawal of immunosuppression or donor lymphocyte infusion, resulting in restoration of donor chimerism and falling FA (figure 1C).
Conclusion - Monitoring of DNMT3A, IDH1, IDH2 and NPM1 mutations in peripheral blood sorted fractions post alloHSCT was feasible and a valuable adjunct to chimerism monitoring. CD34+ sorted peripheral blood samples were most sensitive for early detection of molecular relapse. Serial PB monitoring of recurrent AML mutations, including pre-leukemic clones, may have utility after SCT for AML. Prospective studies of mutational tracking and therapeutic intervention are suggested.
Fleming: Abbvie: Research Funding; Amgen: Honoraria, Membership on an entity's Board of Directors or advisory committees, Research Funding, Speakers Bureau; Astellas: Membership on an entity's Board of Directors or advisory committees; BMS: Speakers Bureau; MSD: Speakers Bureau. Spencer: Amgen: Consultancy, Honoraria, Research Funding; Janssen: Honoraria, Research Funding. Wei: AbbVie, Celgene, Servier: Research Funding; AbbVie, Celgene, Novartis, Amgen, Servier: Membership on an entity's Board of Directors or advisory committees; AbbVie, Celgene, Novartis, Amgen, Servier: Honoraria.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal